Growth hormone doesn't decline because of age. It declines because of this.
Not injections. Not supplements. What your brain actually needs to make growth hormone.
Section 1. Why does growth hormone (GH) dominate longevity conversations?
Growth hormone (GH) is a peptide hormone released in pulses by the pituitary gland, a small endocrine gland located at the base of the brain.
In adults, GH plays several important roles in metabolism and body composition. It helps the body mobilize stored fat for energy (a process called lipolysis), supports the building and maintenance of proteins in tissues such as muscle, and participates in how the body processes carbohydrates and fats. GH also acts on multiple organs—including skeletal muscle, bone, and the liver—both directly and indirectly through another hormone called insulin-like growth factor-1 (IGF-1).
Because GH influences muscle maintenance, body composition, and metabolic regulation, it often appears in discussions about aging and longevity.
Growth hormone (GH) production gradually declines after early adulthood.
Physiological studies show that total daily GH output begins to fall in our 30s largely because the bursts of GH released become smaller.
In longevity circles, that observation is often interpreted in a linear way: GH decreases with age, so perhaps the solution is to increase GH again.
Yet major endocrine reviews and guideline statements have not endorsed raising GH as an anti-aging approach in otherwise healthy adults. Trials in older adults without GH deficiency have produced modest improvements in body composition but also higher rates of adverse effects, and there is no GH-raising therapy approved specifically for anti-aging.
The reason is that GH is not a single dial that can be turned up.
GH output emerges from a regulatory network in the brain involving multiple interacting inputs:
growth hormone–releasing hormone (GHRH), which stimulates GH release
somatostatin, which suppresses GH release
ghrelin, which amplifies GH release
These inputs interact continuously with sleep depth, metabolic state, and reproductive hormones such as testosterone and estrogen.
So when GH changes across the lifespan, the more complete explanation lies in the upstream regulators.
In this article we will look at:
what growth hormone–releasing hormone (GHRH) is and how it stimulates GH production
the lesser-known role of GHRH as a sleep-promoting neuropeptide tied to deep sleep
what tends to change after midlife in the sleep–GHRH–GH axis
whether aging itself is the primary driver of those changes — or whether age is partly a proxy for modifiable factors
Let’s get started.
Section 2. What is growth hormone–releasing hormone (GHRH), and why does it matter more than growth hormone itself?
Growth hormone–releasing hormone (GHRH) is the primary hypothalamic stimulator (a brain input) that promotes the release of growth hormone.
It is produced in a region of the brain called the hypothalamus, specifically in neurons located in the arcuate nucleus. From there, it travels through a specialized vascular network called the hypophyseal-portal circulation—a short blood vessel network that directly connects the hypothalamus and pituitary gland.
When GHRH reaches the pituitary gland, it binds to receptors on specialized cells called somatotrophs, the cells responsible for producing growth hormone.
Binding of GHRH to its receptor activates intracellular signaling pathways—primarily involving cyclic AMP and calcium—that both:
increase GH gene expression and
trigger the release of GH into the bloodstream.
Two under-discussed aspects of GHRH help explain why it plays a central role in GH physiology.
First, GHRH is not only a trigger for moment-to-moment GH release. It also supports the growth and maintenance of somatotroph cells themselves. Animal studies show that when GHRH activity is absent or impaired, the pituitary gland develops with fewer GH-producing cells—a condition known as anterior pituitary hypoplasia.
Second, GHRH production is itself tightly regulated by the body’s internal environment. Inputs related to energy, testosterone, estrogen, and metabolic status all influence how much GHRH is released.
Two of the important inhibitory influences are:
somatostatin, another hypothalamic hormone that directly suppresses GH release
insulin-like growth factor-1 (IGF-1), produced by tissues such as the liver in response to GH
These inputs create a feedback loop. As GH stimulates IGF-1 production in the body, IGF-1 feeds back to the brain to reduce GHRH activity and increase somatostatin, helping regulate future GH pulses.
Physiologically, GHRH therefore acts as part of a three-input architecture.
Somatostatin, produced in the hypothalamus, inhibits GH release and determines when GH pulses are suppressed.
Ghrelin, produced primarily in the stomach and also in the brain, amplifies GH release by activating the GHSR-1a receptor on pituitary cells.
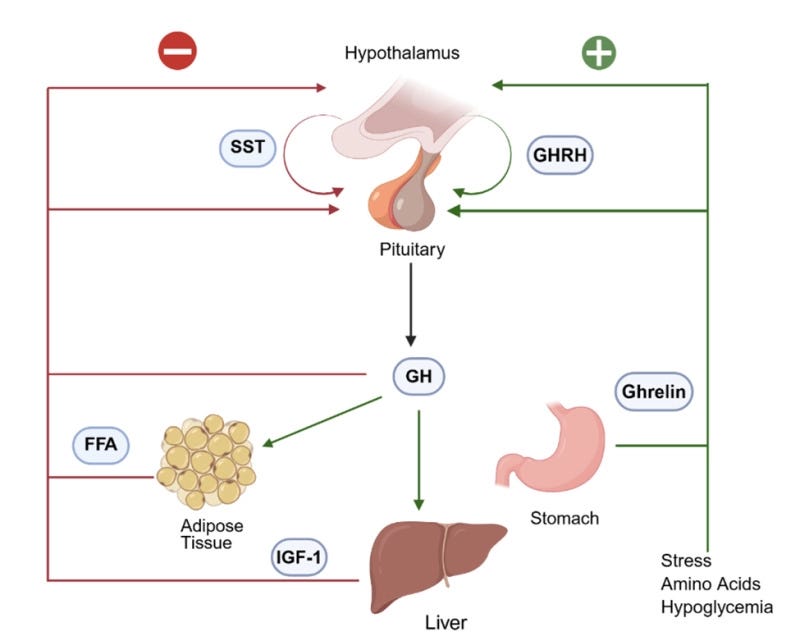
Together these inputs organize GH release into pulses rather than continuous output.
The overall regulatory pattern looks like this:
the hypothalamus releases GHRH, stimulating GH release
the hypothalamus releases somatostatin, suppressing GH release
the pituitary releases GH in pulses
GH stimulates tissues such as the liver to produce IGF-1
IGF-1 feeds back to reduce GHRH and increase somatostatin
This feedback architecture is why GH release remains pulsatile even when researchers experimentally supply GHRH continuously. The alternating hypothalamic inputs—particularly the timing of somatostatin inhibition—organize the pulse pattern.
Why does this matter?
Because GH physiology is organized around pulses rather than steady output.
A large GH pulse delivers a concentrated dose to tissues. Smaller or blunted pulses deliver less of that dose—even if total hormone exposure across the day is not hugely different.
In other words, the physiology depends not only on how much GH exists in the bloodstream, but on the timing and amplitude of the pulses themselves.
And that pattern turns out to be closely connected to sleep.
Section 3. Growth hormone–releasing hormone (GHRH) is also a sleep promoting neuropeptide
One of the more interesting aspects of GHRH physiology is that it does not only regulate growth hormone.
It also participates directly in the regulation of sleep.
Across many species, administering GHRH increases non-REM sleep, particularly the deepest stage known as slow-wave sleep. Conversely, blocking endogenous GHRH activity—using antibodies or antagonists—reduces non-REM sleep and slow-wave activity.
Importantly, this effect does not appear to be explained by GH itself.
In animal studies, impairing GHRH activity produces chronically reduced non-REM sleep that cannot be restored by giving growth hormone. GHRH influences sleep through neural mechanisms that are partly independent of GH.
Experimental work suggests that GHRH can activate sleep-active neurons in brain regions involved in non-REM sleep regulation. When GHRH activity is blocked in these circuits, slow-wave activity decreases.
Human studies generally align with this pattern, although two variables appear to influence the results: how GHRH is delivered and the hormonal context of the individual.
In controlled experiments, episodic intravenous GHRH—given in pulses—tends to increase slow-wave sleep more than continuous infusion.
Average responses also differ somewhat between men and women. In men, peripheral GHRH administration has often been associated with increased slow-wave sleep and higher GH release. Studies including women show more variable effects, suggesting that sex-hormone context influences how the pathway behaves.
This dual role of growth hormone–releasing hormone (GHRH)—regulating both sleep depth and growth hormone (GH) release—helps explain a well-known feature of GH physiology: the largest GH pulse of the day usually occurs shortly after sleep begins.
The sequence often unfolds like this:
sleep onset
the first period of slow-wave sleep
increased hypothalamic GHRH activity and reduced somatostatin restraint
a large pituitary GH pulse
Deep sleep and GH release therefore track together because GHRH sits upstream of both processes.
This relationship raises the next important question.
Section 4. If growth hormone–releasing hormone (GHRH) helps organize both deep sleep and growth hormone (GH) pulses, what happens to this after midlife?
Two hypothalamic changes tend to appear together after midlife:
reduced GHRH drive and/or reduced responsiveness to GHRH
increased somatostatin restraint (somatostatin is the hypothalamic hormone that acts as the brake on GH release)
The downstream result: each GH pulse becomes smaller in amplitude.
This is what endocrinologists sometimes call the somatopause — not a shutdown of growth hormone, but a progressive reduction in pulse size, often cited at ~15% per decade after early adulthood.
Sleep architecture often changes in parallel.
Large meta-analyses of polysomnography recordings (overnight sleep studies that measure brainwaves, breathing, and movement) show that the percentage of slow-wave sleep decreases with age, while lighter sleep stages and nighttime awakenings increase. Because GHRH drives both GH pulses and deep sleep, these changes likely reflect the same hypothalamic changes — and reduced deep sleep, in turn, feeds back to further weaken the GH pulse.
Animal studies provide additional clues about the brain mechanisms involved.
In aged female monkeys, direct sampling at the stalk–median eminence (the junction at the base of the brain where hypothalamic hormones enter the blood supply feeding the pituitary) showed reduced GHRH release along with higher baseline and pulse amplitude of somatostatin release compared with younger animals.
In rodent studies, the number of neurons producing GHRH declines with age, while somatostatin neuron counts also change — suggesting an altered hypothalamic balance between stimulation and inhibition.
Human evidence is necessarily more indirect because hypothalamic peptides cannot easily be measured in living people. Instead, researchers examine GH pulse patterns and responses to experimental GHRH administration.
These studies support the same picture: aging-related reductions in GH release reflect both reduced GHRH drive and stronger somatostatin inhibition.
Taken together, this forms a loop: deep sleep supports GHRH activity, GHRH supports the GH pulse, and the GH pulse supports overnight processes like lipolysis (the breakdown of stored fat for energy) and protein synthesis. When any part of that loop weakens, the downstream output drops.
At first glance, this pattern is often attributed to aging.
But is this purely an aging story? Or is age partly acting as a proxy — a stand-in for factors that accumulate over time and influence sleep depth, GHRH neurons, and the balance between GHRH and somatostatin?
If the decline were purely about age, there wouldn’t be much to work with. But if age is tracking other variables — then those are inputs that can be influenced.
To answer that question, we need to look at what regulates GHRH activity in the first place.
Section 5. What else affects growth hormone–releasing hormone (GHRH) — beyond age?
Age correlates with lower GHRH-linked GH output, but GHRH neurons integrate multiple inputs that change independent of aging.
Several inputs are particularly influential:
sleep pressure and sleep architecture
circadian alignment
reproductive hormones (testosterone and estrogen)
metabolic status and energy balance
Many of these factors can change even before aging becomes a major factor, and they often change alongside it.
To understand how this plays out in practice, we need to examine how these inputs regulate GHRH activity:
1. Sleep pressure and sleep architecture
Slow-wave sleep—the deepest stage of non-REM sleep—is strongly shaped by what sleep researchers call sleep pressure.
Sleep pressure is the body’s built-in drive to sleep. The longer someone stays awake, the more this pressure builds. When sleep finally begins, that accumulated pressure expresses itself as deeper slow-wave sleep and larger slow-wave brain activity.
This matters for growth hormone because the largest GH pulses occur during deep non-REM sleep.
When slow-wave sleep is strong, the conditions are more favorable for a large early-night GH pulse
When sleep becomes fragmented—or when deep sleep becomes harder to generate—the GH pulse that normally accompanies it tends to become smaller.
Studies across both humans and animals show the same pattern. Sleep deprivation blunts the normal nocturnal GH surge, while recovery sleep—when deep sleep rebounds—often produces a stronger sleep-related GH pulse
2. Circadian alignment: when you sleep matters — even if how long you sleep doesn’t change
Sleep pressure determines how strongly the brain wants sleep, but circadian timing determines when the body expects sleep to occur.
The circadian rhythm is orchestrated by a small cluster of neurons in the hypothalamus called the suprachiasmatic nucleus (SCN). This structure acts as the brain’s internal clock, coordinating daily rhythms in sleep, body temperature, metabolism, and hormone release.
GH release is primarily linked to sleep onset and slow-wave sleep, but the circadian system modulates the hormonal environment in which that sleep-related pulse occurs.
Although sleep itself is the primary trigger for the large nightly GH pulse, the circadian clock modulates the hormonal environment in which that pulse occurs.
Classic studies of the sleep–GH relationship show that the first GH surge after sleep onset coincides with a period in which hypothalamic GHRH activity rises while somatostatin inhibition temporarily relaxes. The circadian clock helps create a window during which the sleep-triggered pulse can occur.
When sleep timing becomes misaligned with the internal clock—through irregular schedules or chronic circadian disruption—the hormonal environment that normally supports this pulse can weaken.
Circadian misalignment also influences glucose regulation and insulin sensitivity, both of which interact with the GH axis through hypothalamic and pituitary pathways. Hello
So even when sleep duration is unchanged, when sleep occurs relative to the internal clock can influence how endocrine rhythms behave.
3. How testosterone and estrogen shape the growth hormone (GH) axis across the lifespan
Sex hormones also shape the growth hormone (GH) axis.
The best-documented example occurs during puberty, when rising sex-steroid levels are associated with a large increase in GH release. Much of this effect appears to come from increased pulse frequency and pulse amplitude.
In adult physiology, testosterone and estrogen influence the axis in somewhat different ways.
Androgens, including testosterone, influence both GH secretion and the peripheral actions of GH.
Estrogens are described as stimulating GH release while also altering how GH acts through the liver and other tissues. Experimental studies show that estrogen exposure can increase pituitary responsiveness to GHRH and reduce somatostatin inhibition.
Across midlife, the sex-hormone milieu often changes.
In men, testosterone levels and androgen signaling often decline or become more easily disrupted with age. In women, the transition through perimenopause and menopause produces more pronounced changes in estrogen and progesterone patterns.
Because these hormones interact with hypothalamic regulation, pituitary responsiveness, and sleep architecture, changes in the sex-steroid environment can influence GH pulsatility through multiple pathways.
4. How metabolic health influences the growth hormone (GH) axis
Energy balance is also an important regulator of the growth hormone (GH) axis.
Many of these effects act at the hypothalamic level—through hypothalamic regulation and pituitary responsiveness—not only through the pituitary gland’s capacity to produce GH.
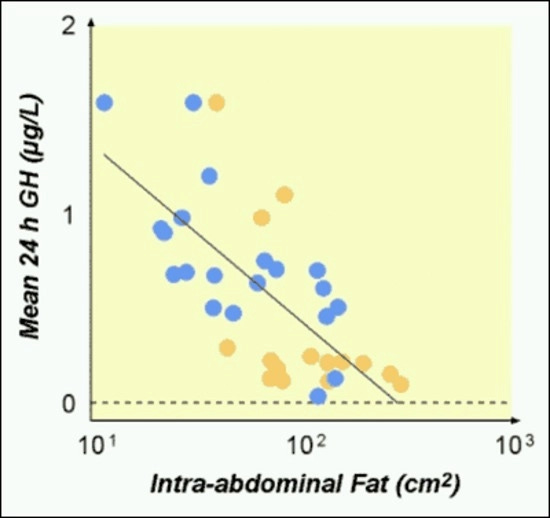
Several metabolic patterns commonly observed in midlife illustrate this relationship.
Visceral fat—the fat stored around abdominal organs—is consistently associated with lower GH release.
In obesity, basal and pulsatile GH secretion tend to be reduced, while pulse frequency is often relatively preserved.
Insulin and circulating fatty acids also influence the axis.
High insulin levels can suppress GH release. In experimental settings, GH output can decrease within days of overfeeding as insulin rises. Cell-culture and animal studies also show that insulin can reduce the expression of genes involved in GH production and release.
Free fatty acids exert a similar effect. Elevated fatty acid levels inhibit GH release, while lowering fatty acids—such as by temporarily suppressing fat breakdown—can increase GH responses during stimulation testing.
IGF-1 adds another dimension. IGF-1 is the hormone produced by tissues in response to GH, and it feeds back to the brain and pituitary to reduce GH drive through negative feedback.
In obesity, total IGF-1 often does not decline in parallel with GH, and free IGF-1 can even be higher, partly because insulin and IGF-binding proteins shift how much IGF-1 is bioavailable. This feedback can further suppress the hypothalamic inputs that drive GH pulses.
Taken together, several physiological changes that often appear with age—sleep fragmentation, increased visceral fat, higher insulin exposure, and altered testosterone and estrogen signaling—can all reshape the balance between GHRH stimulation and somatostatin inhibition.
From that perspective, chronological age often travels alongside these changes rather than acting as the only driver.
Section 6. Growth hormone (GH) gets the attention — but the factors that determine whether the nightly pulse still happens are within reach
Growth hormone tends to dominate longevity conversations, but it sits downstream in the physiology.
The question that matters is whether the conditions that allow a strong GH pulse are still present.
Each of the inputs we looked at — sleep depth, circadian timing, metabolic state, the sex-hormone environment — can change independently of the calendar. And each one feeds into the GHRH–somatostatin balance that determines pulse size.
Midlife GH decline often reflects the cumulative effect of several physiological inputs that change over time — many of which can be influenced.
None of those inputs ensure a larger GH pulse.
But they do shape the physiology that determines whether the pulse has the opportunity to occur.
And that opens a more constructive question for midlife and later life:
How can we support the conditions — sleep architecture, circadian alignment, metabolic health, and the hormonal environment — so that the brain and endocrine axis can continue running the nightly repair programs they were built to run?
When those conditions are supported, the physiology doesn’t need to be forced. Deep sleep, GHRH activity, and GH pulsatility can continue to function more favorably across midlife and later life — coordinating overnight restoration, maintenance, and metabolic regulation.
That’s the part of the growth hormone conversation that rarely gets discussed. And it’s within your influence.
—Kat
P.S. If your sleep feels less restorative than it used to — and you’re wondering which part of the equation is involved — my free 3AM Decoder maps 3am waking patterns across 5 patterns. It takes about 2 minutes:
References
Cappola, A. R., Auchus, R. J., El-Hajj Fuleihan, G., Handelsman, D. J., Kalyani, R. R., McClung, M., Stuenkel, C. A., Thorner, M. O., & Verbalis, J. G. (2023). Hormones and aging: An Endocrine Society scientific statement. The Journal of Clinical Endocrinology & Metabolism, 108(8), 1835–1874.
U.S. Food and Drug Administration. (2020, September 1). FDA approves weekly therapy for adult growth hormone deficiency.
Olarescu, N. C., Gunawardane, K., Hanson, T. K., Møller, N., & Jørgensen, J. O. L. (2025, April 18). Normal physiology of growth hormone in normal adults. In Endotext [Internet]
Fredrick, J. R., Blackman, M. R., Corpas, E., Merriam, G. R., Kargi, A. Y., & Garcia, J. M. (2026, February 13). Growth hormone and aging. In Endotext [Internet].
Stojilkovic, S. S. (2012). Molecular mechanisms of pituitary endocrine cell calcium handling. Cell Calcium, 51(3–4), 212–221.
Mayo, K. E., Miller, T., DeAlmeida, V., Godfrey, P., Zheng, J., & Cunha, S. R. (2000). Regulation of the pituitary somatotroph cell by GHRH and its receptor. Recent Progress in Hormone Research, 55, 237–266.
Frohman, L. A., & Kineman, R. D. (2002). Growth hormone-releasing hormone and pituitary somatotrope proliferation. Minerva Endocrinologica, 27(4), 277–285.
Obál, F., Jr., Kapás, L., Gardi, J., Taishi, P., Bodosi, B., & Krueger, J. M. (1999). Insulin-like growth factor-1 (IGF-1)-induced inhibition of growth hormone secretion is associated with sleep suppression. Brain Research, 818(2), 267–274.
Hulse, J. A., Rosenthal, S. M., Cuttler, L., Kaplan, S. L., & Grumbach, M. M. (1986). The effect of pulsatile administration, continuous infusion, and diurnal variation on the growth hormone (GH) response to GH-releasing hormone in normal men. The Journal of Clinical Endocrinology & Metabolism, 63(4), 872–878.
Obal, F., Jr., & Krueger, J. M. (2004). GHRH and sleep. Sleep Medicine Reviews, 8(5), 367–377.
Marshall, L., Mölle, M., Böschen, G., Steiger, A., Fehm, H. L., & Born, J. (1996). Greater efficacy of episodic than continuous growth hormone-releasing hormone (GHRH) administration in promoting slow-wave sleep (SWS). The Journal of Clinical Endocrinology & Metabolism, 81(3), 1009–1013.
Van Cauter, E., & Plat, L. (1996). Physiology of growth hormone secretion during sleep. The Journal of Pediatrics, 128(5 Pt 2), S32–S37.
Russell-Aulet, M., Dimaraki, E. V., Jaffe, C. A., DeMott-Friberg, R., & Barkan, A. L. (2001). Aging-related growth hormone (GH) decrease is a selective hypothalamic GH-releasing hormone pulse amplitude mediated phenomenon. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 56(2), M124–M129.
Ohayon, M. M., Carskadon, M. A., Guilleminault, C., & Vitiello, M. V. (2004). Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: Developing normative sleep values across the human lifespan. Sleep, 27(7), 1255–1273.
Brandenberger, G., Gronfier, C., Chapotot, F., Simon, C., & Piquard, F. (2000). Effect of sleep deprivation on overall 24 h growth-hormone secretion. The Lancet, 356(9239), 1408. h
Clasey, J. L., Weltman, A., Patrie, J., Weltman, J. Y., Pezzoli, S., Bouchard, C., Thorner, M. O., & Hartman, M. L. (2001). Abdominal visceral fat and fasting insulin are important predictors of 24-hour GH release independent of age, gender, and other physiological factors. The Journal of Clinical Endocrinology & Metabolism, 86(8), 3845–3852.
Morris, C. J., Aeschbach, D., & Scheer, F. A. J. L. (2012). Circadian system, sleep and endocrinology. Molecular and Cellular Endocrinology, 349(1), 91–104.
Mauras, N., Blizzard, R. M., Link, K., Johnson, M. L., Rogol, A. D., & Veldhuis, J. D. (1987). Augmentation of growth hormone secretion during puberty: Evidence for a pulse amplitude-modulated phenomenon. The Journal of Clinical Endocrinology & Metabolism, 64(3), 596–601.