Is the gut-sleep link finally real, or wellness hype?
Gut GABA and sleep: what the 2026 data shows & what it means for personalized sleep solutions
This article part II of the collaboration with Dr. Mike Hunter.
Dr. Mike Hunter is a retired physician specializing in Internal Medicine and Gastroenterology, with over 20 years in academic medicine. He has headed medical units and the Gastroenterology Department at Helen Joseph Hospital, University of the Witwatersrand, South Africa, and practised in the NHS in the UK. Mike has lectured at all levels, published internationally, and now write medical articles focusing on the gut microbiome, mitochondrial health, exercise, mental and cardiovascular wellness, photobiomodulation, and circadian rhythms.
What if your gut was influencing how well you sleep each night?
GABA — gamma-aminobutyric acid — is one of the brain’s main calming molecules, and many sleep medicines work by boosting its effects. Gut microbes can also make GABA. But whether that gut-made GABA reaches the brain, and whether it matters for sleep, is a question the field is only now beginning to answer with human data.
GABA in blood is thought to reach the brain in only small amounts, if at all, under typical conditions — and human evidence about GABA crossing the blood–brain barrier is currently limited. Few human studies measure gut GABA and sleep at the same time, and even fewer also measure brain GABA.
So does gut-produced GABA affect human sleep — and if so, how?
This review will discuss:
What is gut-produced GABA, and how do microbes make it?
What pathways exist for gut-made GABA to affect human sleep (even if it does not cross the blood-brain-barrier)?
Human evidence linking gut GABA to sleep
Practical implications: is optimizing gut-made GABA a practical strategy for improving your sleep?
Let’s get started.
What is gut-produced GABA, and how do microbes make it?
GABA is a chemical messenger that usually lowers brain cell “firing.” In the brain, GABA helps support sleep and calming.
Many gut microbes can make (or break down) neuroactive chemicals, including GABA.
The main microbial pathway: glutamate → GABA
The most common microbial route is:
Glutamate (or glutamine → glutamate) → GABA, using glutamate decarboxylase (GAD). This enzyme needs pyridoxal-5′-phosphate (PLP) (a vitamin B6 form) to work.
Many bacteria also use a transporter (often described as a glutamate/GABA antiporter, sometimes linked to “gadC”) that helps move glutamate in and GABA out. This pathway is often tied to surviving acidic conditions, because turning glutamate into GABA uses up a proton and can help the microbe handle low pH.
Which gut microbes can make GABA?
GABA production is highly strain-dependent (two microbes in the same genus can act differently). Still, several common gut groups include GABA producers:
Bacteroides: A large study of human intestinal isolates found GABA-making genes were very common in this genus, and most tested strains made GABA in lab conditions.
Lactobacillus/Lactiplantibacillus and Bifidobacterium: These groups include well-known GABA producers (often used in foods or supplements).
One study focused on gut Bacteroides found:
About 90% of Bacteroides genomes (and 96% of human gut Bacteroides genomes) had the full gene set for the GABA pathway they studied.
In lab testing, 16 out of 17 human intestinal Bacteroides strains made GABA, ranging from 0.09 to 60.84 mM (a wide range).
Production depended on precursor supply (glutamate/glutamine) and pH.
Production is not the same as “net GABA level”
Even if many microbes can make GABA, the measured GABA in stool depends on:
how much producers make,
how much other microbes use up (some microbes can consume GABA as a fuel), and
how fast GABA is absorbed or broken down by the host.
For example, one gut microbe highlighted in a major paper used GABA as a carbon/energy source, showing that “GABA consumers” exist in the gut community.
Does gut-made GABA affect sleep?
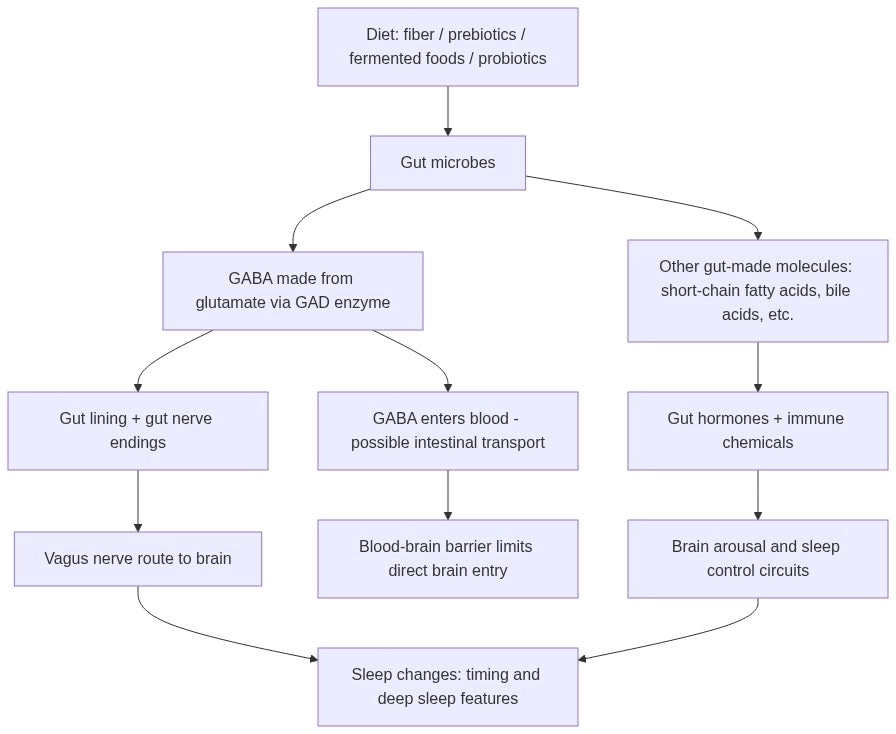
A key idea: gut-made GABA is made in the gut lumen, while sleep is mainly controlled by brain circuits. So the question is: how could gut GABA influence the brain parts that control sleep?
Research points to several routes that can work together.
1. Route through gut nerves and the vagus nerve
The gut has a large nerve network. Some of its nerve endings connect to the brain using the vagus nerve.
One strong animal study showed that feeding a certain Lactobacillus strain changed brain GABA receptor gene expression and stress-related behavior in mice, and these effects depended on the vagus nerve (they went away when the vagus nerve was cut). This supports a gut→vagus→brain route for microbe-linked GABA effects (at least in animals).
Human trials reviewed below do not directly prove vagus nerve steps, but some do show that gut interventions that raise stool or urine GABA also change sleep timing and deep sleep features, which is consistent with a nerve-mediated route.
2. Route through immune and inflammation pathways
Sleep and inflammation are linked. Gut microbes can change immune chemicals (like cytokines), and those can affect sleep regulation in the brain.
Several insomnia-related human microbiome studies focus on inflammation and blood metabolites as links between the gut and insomnia symptoms. While these studies do not always measure GABA directly, they support the idea that gut changes can affect sleep through immune-related routes.
GABA also has receptors outside the brain, including on some gut and immune cell types, so it may influence immune activity and gut function locally, which can then feed into sleep regulation. (Much of this part is still better supported in lab/animal studies than in humans.)
3. Route through metabolites and gut hormones
Gut microbes make many small molecules that can affect brain function even when those molecules are not classic neurotransmitters.
A strong example in humans is a prebiotic yeast mannan trial: the authors found that changes in fecal propionate (a short-chain fatty acid) statistically explained part of the increase in time in bed, and changes in fecal GABA statistically explained part of the improvement in deep-sleep (N3) latency.
This matters because it suggests:
GABA may be one route, but
it may work alongside other gut-made molecules (like short-chain fatty acids) that can affect gut hormones and brain arousal.
4. Blood–brain barrier considerations: does gut GABA reach the brain?
A long-standing concern is that GABA is thought to cross the blood–brain barrier poorly, which raises a puzzle: if you raise GABA in the gut or blood, how does the brain “feel” it?
A major review on GABA as a supplement summarizes that many researchers have considered BBB crossing minimal, and it highlights uncertainty about how oral GABA could affect brain outcomes.
A more recent review in a neuroscience journal also notes mixed evidence about BBB permeability for GABA, and it emphasizes that the BBB issue is a big reason researchers look at nerve or gut-mediated routes instead of direct brain entry.
Separately, cell studies suggest gut cells can transport GABA across intestinal cell layers via carrier-mediated transport, which could help GABA enter the bloodstream.
Putting these together, the sensible human-centered takeaway is:
Gut interventions can raise GABA measured in stool or urine (and sometimes blood),
sleep outcomes can shift,
but direct proof that gut-made GABA itself enters the brain and raises brain GABA in humans is still limited.
Human evidence linking gut GABA to sleep
This section prioritizes human studies that connect (1) a gut intervention, (2) a GABA measure (stool, urine, or blood), and (3) sleep outcomes.
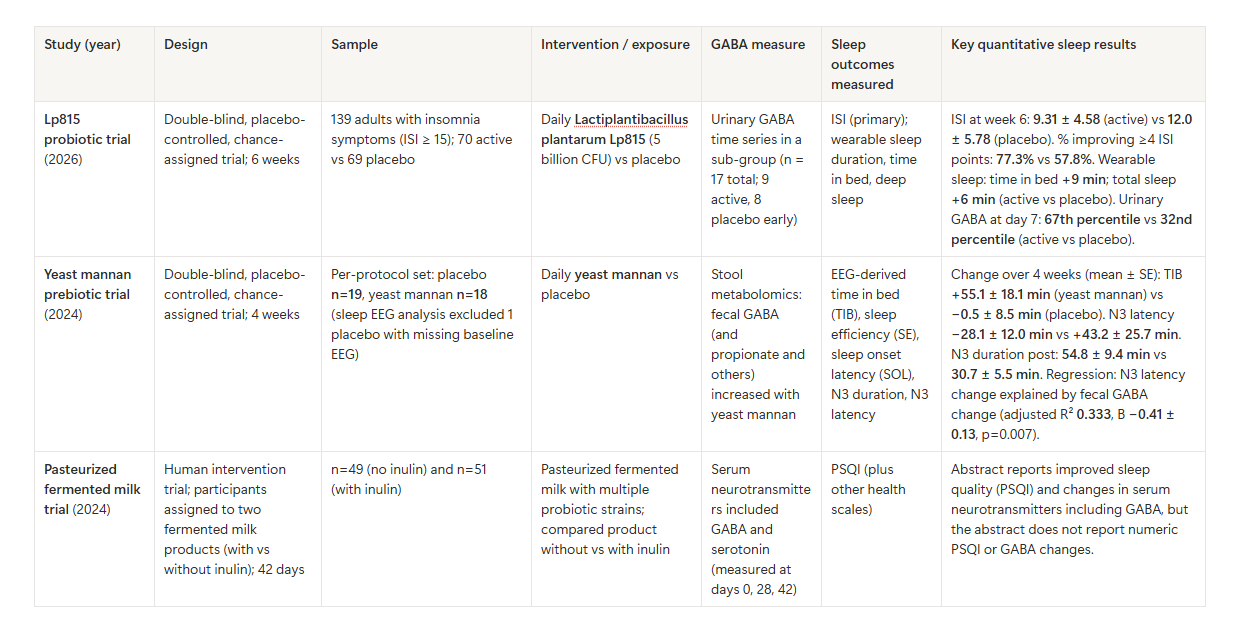
Key human trials with a GABA measure and sleep outcomes
The table below compares the most relevant human trials found that measured GABA (or a close proxy) and sleep.
Across these trials, there is a pattern:
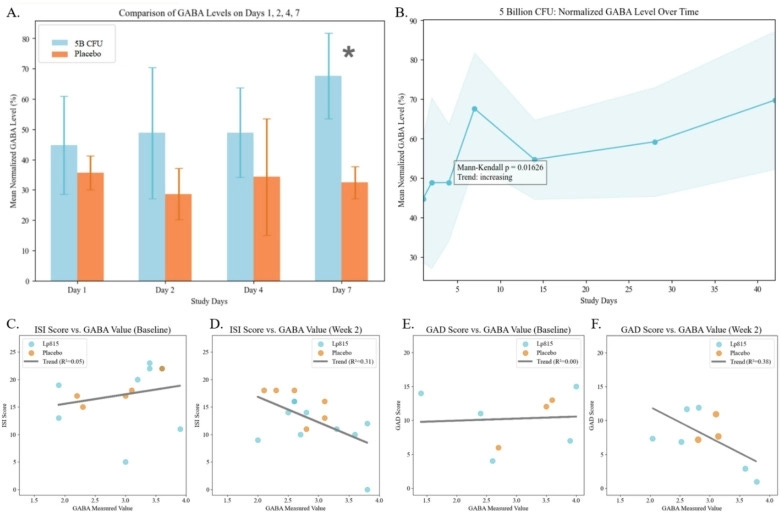
A gut-targeting intervention increases a GABA-related measure (stool or urine, and possibly blood).
Some sleep measures improve, especially insomnia symptom scores (ISI) and deep sleep timing (N3 latency) in the yeast mannan EEG study.
The strongest “gut GABA → sleep” link within one trial is in the yeast mannan study’s regression analysis, where fecal GABA change was tied to N3 latency change.
At the same time, the probiotic Lp815 trial shows only small mean differences in wearable-measured sleep duration (minutes), even though the insomnia symptom score improved more noticeably. That can happen because insomnia questionnaires measure distress and perceived sleep problems, not just minutes asleep.
Because human mechanistic work is still thin, animal data is used to explain “how it might work.”
Two examples that matter for:
The vagus nerve requirement for a Lactobacillus strain’s effect on brain GABA receptor expression (mouse).
GABA-rich fermented milk improving sleep behavior in mice, alongside gut microbiome and short-chain fatty acid changes.
These do not prove human effects, but they help explain why researchers focus on nerve and metabolite routes from gut to brain.
Limitations and gaps: Few human studies directly connect gut GABA and sleep
Most human sleep–microbiome studies do one of these, but not all at once:
measure sleep plus gut microbes, but not GABA, or
measure stool or blood metabolites but not sleep in detail, or
test a gut intervention and measure sleep, but do not measure GABA.
The trials above stand out because they include both GABA measures and sleep outcomes, but the list is still short.
Why GABA may not be acting alone
Gut interventions change many things at the same time:
microbial mix,
short-chain fatty acids,
bile acids,
gut motility and comfort,
immune chemicals, and
many other metabolites.
In the yeast mannan study, propionate and GABA both tied to different sleep variables, suggesting “many molecules, many routes.”
In the Lp815 probiotic study, urinary GABA rose early (in a small sub-group), while wearable sleep changes were modest in minutes. That could mean GABA acts more on sleep quality feelings, stress, or arousal, and less on total minutes asleep—or it could reflect measurement limits.
Stool GABA is not the same as gut lumen GABA at nerve endings. Stool is what comes out; it mixes microbes, diet residues, and host secretions.
Urinary GABA is an indirect marker. It reflects what ended up in urine after filtration and metabolism. In the Lp815 trial, urine sampling was done in only 17 participants total, and placebo urine data later in the study was sparse.
Wearables and simplified EEG are helpful but not the same as full sleep lab testing. The Lp815 study used a wearable ring for sleep staging, and the yeast mannan study used a patch-type EEG device. Both are useful, but they do not capture as much as full polysomnography with multiple channels.
Blood–brain barrier uncertainty remains a key gap
Even when gut interventions raise GABA in stool or urine (and sometimes blood), it remains uncertain how much of that GABA can reach the brain directly in typical human conditions. Many studies still view nerve and gut-mediated routes as more likely than direct BBB crossing, but more human data is needed.
Practical implications: What seems reasonable based on the human data so far
Gut-targeting approaches may help some people with insomnia symptoms, but effects vary and may be modest for “minutes asleep.” The Lp815 trial showed a moderate-sized improvement in insomnia symptom score (ISI) and small increases in wearable-measured sleep time.
Prebiotics may affect deep sleep timing. In the yeast mannan trial, N3 latency improved (shortened) and time in bed increased, and fecal GABA change tied to N3 latency change.
Fermented foods or fermented milk products may influence GABA-related markers and sleep quality, but it can be hard to separate “GABA consumed in food” vs “GABA made by gut microbes after the food changes the gut.” The pasteurized fermented milk trial reported serum neurotransmitter changes (including GABA) along with PSQI improvement, but detailed numbers are needed to judge effect size.
If the goal is “more gut-made GABA,” what does the research suggest?
Based on the mechanisms and trials above:
Strain matters. “Probiotic” is not one thing; GABA production varies widely by strain and gut conditions (like pH and glutamate availability).
Food feeds the microbes. Prebiotics (like yeast mannan) can shift microbial metabolism and raise fecal GABA and other metabolites.
Net GABA depends on both producers and consumers. Raising “GABA producers” does not guarantee higher stool GABA if other microbes use it.
In sum, human research supports a possible link between gut interventions, gut-associated GABA measures (stool or urine), and some sleep outcomes. The most direct human evidence includes: a probiotic trial showing improved insomnia symptom scores plus higher urinary GABA (in a small urine sub-group) and a prebiotic trial showing improved deep sleep timing plus higher fecal GABA.
What is still missing is large, independent human trials that measure gut GABA + blood GABA + brain GABA + gold-standard sleep testing in the same people, so the field can pin down which route matters most.
Who may benefit from targeting gut-derived GABA for sleep
Gut-derived GABA is not a primary approach for insomnia, and it is not a substitute for getting the fundamentals right — consistent sleep timing, adequate sleep opportunity, managing stress and inflammation. Those remain where the largest gains come from.
But there is a specific group where gut-derived GABA may matter: those who are tired but wired. People whose sleep is shaped more by hyperarousal — elevated sympathetic tone, difficulty reducing arousal at night — than by lack of opportunity.
Why this group? Because hyperarousal involves the same pathways that gut-brain communication can influence: vagal tone, inflammatory markers, and neurotransmitter balance. The Lp815 trial participants had insomnia shaped in part by anxiety and arousal, and they showed the largest improvements in perceived sleep quality. The yeast mannan trial showed changes in deep sleep onset timing — the sleep stage most sensitive to arousal state.
For someone in this group, targeting the gut microbiome through diet (fermented foods, prebiotic fiber), prebiotics (like yeast mannan), or carefully selected probiotic strains (with evidence for GABA production in vivo, not just in lab conditions) may offer a modest but meaningful change in sleep quality.
The effect is unlikely to be fast.
But it is physiologically sensible and likely will benefit sustainable root-cause centered sleep improvement strategies, supported by early human data, and consistent with what the gut-brain communication routes described in the literature.
—Kat
P.S. If you are waking around 3am, stuck at a 5-6 hour sleep ceiling, or still tired after the usual sleep strategies, I am opening VIP early access for personalized sleep intelligence (currently exclusively for men 40+), based on your sleep pattern, sleep quality, and daytime activity. Join the waitlist (or share this with someone you care about who might benefit from it).
Personalized sleep intelligence, tailored to the complexity of men’s 40+ physiology (coming to iOS & Android):
References
Grant, A. D., Erfe, M. C. B., Delebecque, C. J., Keller, D., Zimmerman, N. P., Kazaryan, A., Oliver, P. L., Moos, J., Luna, V., & Craft, N. (2026). Lactiplantibacillus plantarum Lp815 improves sleep and increases urinary GABA in a randomized, double-blind, placebo-controlled study of sleep disturbance. Scientific Reports, 16, Article 644.
Tanihiro, R., Yuki, M., Sasai, M., Haseda, A., Kagami-Katsuyama, H., Hirota, T., Honma, N., & Nishihira, J. (2023). Effects of prebiotic yeast mannan on gut health and sleep quality in healthy adults: A randomized, double-blind, placebo-controlled study. Nutrients, 16(1), Article 141.
Braga, J. D., Thongngam, M., & Kumrungsee, T. (2024). Gamma-aminobutyric acid as a potential postbiotic mediator in the gut-brain axis. npj Science of Food, 8(1), Article 16.
Otaru, N., Ye, K., Mujezinovic, D., Berchtold, L., Constancias, F., Cornejo, F. A., Krzystek, A., de Wouters, T., Braegger, C., Lacroix, C., & Pugin, B. (2021). GABA production by human intestinal Bacteroides spp.: Prevalence, regulation, and role in acid stress tolerance. Frontiers in Microbiology, 12, Article 656895.
Strandwitz, P., Kim, K. H., Terekhova, D., Liu, J. K., Sharma, A., Levering, J., McDonald, D., Dietrich, D., Ramadhar, T. R., Lekbua, A., Mroue, N., Liston, C., Stewart, E. J., Dubin, M. J., Zengler, K., Knight, R., Gilbert, J. A., Clardy, J., & Lewis, K. (2019). GABA-modulating bacteria of the human gut microbiota. Nature Microbiology, 4(3), 396–403.
Boonstra, E., de Kleijn, R., Colzato, L. S., Alkemade, A., Forstmann, B. U., & Nieuwenhuis, S. (2015). Neurotransmitters as food supplements: The effects of GABA on brain and behavior. Frontiers in Psychology, 6, Article 1520.
Hepsomali, P., Groeger, J. A., Nishihira, J., & Scholey, A. (2020). Effects of oral gamma-aminobutyric acid (GABA) administration on stress and sleep in humans: A systematic review. Frontiers in Neuroscience, 14, Article 923.
Zhou, J., Wu, X., Li, Z., Zou, Z., Dou, S., Li, G., Yan, F., Chen, B., & Li, Y. (2022). Alterations in gut microbiota are correlated with serum metabolites in patients with insomnia disorder. Frontiers in Cellular and Infection Microbiology, 12, Article 722662.
Murakami, H., Ko, T., Ouchi, H., Namba, T., Ebihara, S., & Kobayashi, S. (2024). Bifidobacterium adolescentis SBT2786 improves sleep quality in Japanese adults with relatively high levels of stress: A randomized, double-blind, placebo-controlled study. Nutrients, 16(11), Article 1702.
Bravo, J. A., Forsythe, P., Chew, M. V., Escaravage, E., Savignac, H. M., Dinan, T. G., Bienenstock, J., & Cryan, J. F. (2011). Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proceedings of the National Academy of Sciences of the United States of America, 108(38), 16050–16055.
Yu, L., Han, X., Cen, S., Duan, H., Feng, S., Xue, Y., Tian, F., Zhao, J., Zhang, H., Zhai, Q., & Chen, W. (2020). Beneficial effect of GABA-rich fermented milk on insomnia involving regulation of gut microbiota. Microbiological Research, 233, Article 126409.
Zhao, J., Li, H., Kwok, L.-Y., Wu, X., Liu, H., Shen, X., Zhao, F., Qi, H., Ma, H., & Sun, Z. (2024). Improvement of sleep quality and sub-health conditions through pasteurized fermented milk consumption: A human intervention study. Journal of Functional Foods, 122, Article 106562.




I love this! Two of my favorite writers in one place. What I found most useful is the idea that the gut-sleep connection may matter most for people who feel tired but wired. In other words, the issue may not simply be needing more sleep, but having a body and nervous system that cannot fully calm down at night. You do a good job showing how the gut may play a role in that process through inflammation, stress, vagal tone, and the gut-brain connection, without pretending that one supplement or one pathway explains it all. Few things in life ever are.