Can 3–5 a.m. wakeups affect cancer risk?
Sleep touches nearly every process that matters for cancer biology.
Once we pass midlife, our baseline cancer risk goes up simply because mutations, immune aging, and long-term exposures have had more time to accumulate.
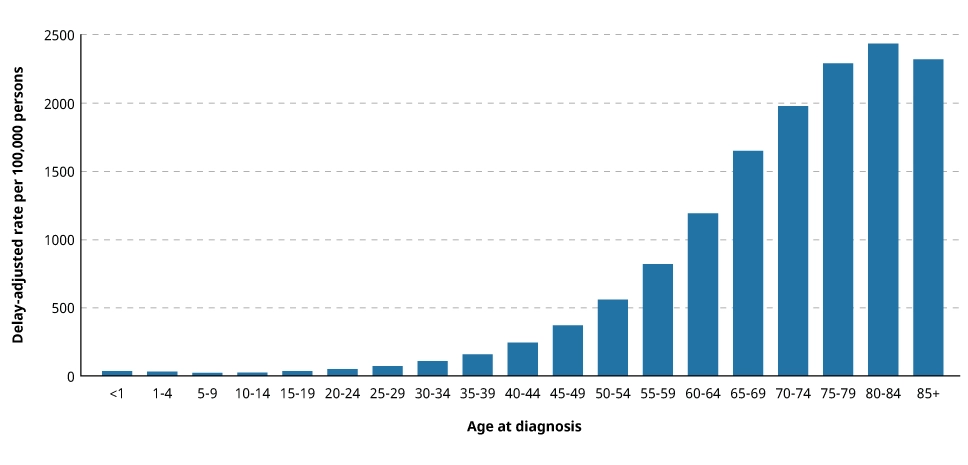
Advancing age is the most important risk factor for cancer overall and for many individual cancer types. The incidence rates for cancer overall climb steadily as age increases, from < 26 cases per 100,000 people in age groups < age 20, to about 350 per 100,000 people among those aged 45–49, to more than 1,000 per 100,000 people in age groups 60 years+—National Cancer Institute
We can’t change our genetics or our early-life environment, but we can still influence several day-to-day inputs that shape cancer risk over the coming years.
Sleep touches nearly every process that matters for cancer biology:
immune surveillance,
DNA repair,
hormone balance (especially sex hormones),
insulin sensitivity, and
the coordination of cell division with the light–dark cycle.
When that coordination breaks down, cells can begin dividing at the wrong times, under hormonal and inflammatory conditions that favor tumor growth.
For someone past midlife, the practical question becomes: if you invest effort into getting “good enough” sleep — adequate duration, decent quality, and reasonably stable timing in a dark nighttime environment — does that move the needle on cancer risk and outcomes?
In this article, we’ll cover:
A 2026 review linking sleep traits (chronotype, insomnia) to hormone-sensitive cancer risk.
What a 2025 U.S. survivor cohort tells you about sleep duration and cancer mortality
Whether sleep duration is linked to first-time cancer diagnoses in midlife adults without cancer at baseline
What a light-tumor experiment in rats reveals about light at nighttime, circadian rhythms, and tumor growth.
Five practical sleep-and-circadian moves you can use
Let’s get started.
Study 1: Sleep traits and hormone-sensitive cancers – insights from a 2026 review
In a recent February 2026 review —investigators combined 62 observational studies (45 cohort, 17 case-control) looking at sleep traits and reproductive cancers (breast, prostate, endometrial, epithelial ovarian), with data drawn largely from middle-aged and older adults.
They focused on four modifiable traits:
usual sleep duration,
insomnia,
chronotype (whether someone is more “morning-type” or “evening-type”), and
snoring.
Key findings for reproductive cancer risk:
Evening chronotype was linked to higher breast cancer risk compared with morning chronotype: HR = 1.12 (95% CI 1.07–1.17; I² = 14%; 4 cohorts), ~12% higher risk.
Evening chronotype also carried higher epithelial ovarian cancer risk: HR = 1.15 (95% CI 1.02–1.29; I² = 0%; 2 cohorts), ~15% higher risk.
Insomnia symptoms were associated with elevated breast cancer risk in crude models (RR = 1.23; 95% CI 1.01–1.50; I² = 94%; 9 cohorts), and diagnosed insomnia had a similar magnitude (RR = 1.26; 95% CI 1.01–1.58; I² = 96%; 5 studies).
When they restricted to studies with more thorough adjustment (e.g., for depression, lifestyle, and comorbidities), the insomnia–breast cancer association shrank and lost statistical significance (RR = 1.04; 95% CI 0.95–1.14; I² = 41.5%; 3 cohorts).
Overall, the authors concluded that the chronotype findings, with low heterogeneity, give the support for circadian disruption as a contributor to reproductive cancer risk.
Taken together, this review suggests that being strongly evening-type and living in ways that reinforce that (late bedtimes, late meals, more light at nighttime) may carry a modest but measurable elevation in hormone-sensitive cancer risk.
The insomnia story is weaker once you adjust for mood and lifestyle, which reminds you that poor sleep often travels with other risk factors.
The practical meaning is not necessarily that you must “become a morning person”, but that nudging your schedule earlier and making your sleep timing more aligned with the light–dark cycle may help on the margin. Most of the underlying cohorts adjusted for age, BMI, smoking, alcohol, and sometimes hormone therapy and reproductive history, so the chronotype associations are not just a proxy for those classic factors.
Study 2: Sleep duration and cancer survival – 2025 NHANES survivors analysis
A recent 2025 analysis— using NHANES data—looked at 2,528 U.S. adults who reported a prior cancer diagnosis, with an average age of about 65 years and follow-up for mortality out to the end of 2019.
Sleep duration came from self-report and was grouped into short (<6 hours per nighttime), normal (6–8 hours), and long (>8 hours). They also calculated weekly moderate-to-vigorous physical activity (MVPA) using the Global Physical Activity Questionnaire and classified people as either meeting MVPA recommendations (≥600 MET-minutes/week) or not.
Key findings for mortality in cancer survivors:
Both short and long sleep durations were associated with higher all-cause mortality vs 6–8 hours:
Short sleep: HR = 1.382 (95% CI 1.377–1.389).
Long sleep: HR = 1.290 (95% CI 1.286–1.295).
For cancer-specific mortality, extremes of sleep were again unfavorable:
Short sleep: HR = 1.593 (95% CI 1.584–1.602).
Long sleep: HR = 1.478 (95% CI 1.470–1.486).
Physically active survivors had substantially lower mortality: HR = 0.486 (95% CI 0.484–0.488) for cancer mortality compared with inactive peers.
The combination of extreme sleep duration and low activity was worst: survivors with both poor MVPA and short sleep had HR = 1.554 (95% CI 1.548–1.561) for all-cause mortality, and those with poor MVPA and long sleep had HR = 1.674 (95% CI 1.668–1.680).
Among survivors who met MVPA targets, the increased mortality risk associated with extreme sleep durations dropped noticeably; some combinations had HRs below 1.0.
This analysis suggests that for people already living with or beyond cancer, sleeping in the mid-range (about 6–8 hours) is associated with better survival, especially when you combine that with regular movement. The main multivariable model adjusted for age, sex, race, BMI, education levels, smoking, drinking, sleep disorder and general health condition defined as history of diabetes, hypertension and CVD.
Study 3: Sleep duration and first-time cancer diagnoses –adults 45+
In a 2024 prospective cohort—researchers followed 9,996 adults aged 45 and older who had no cancer at baseline, across about 88,790 person-years of observation.
Total daily sleep duration was defined as nocturnal sleep plus daytime naps and categorized as short (≤6 hours per 24 hours), moderate (6–9 hours), or long (>9 hours).
Key findings for incident cancer:
Over a median 9-year follow-up, 325 participants developed cancer (incidence rate ≈ 20.5 per 1,000 person-years).
Compared with 6–9 hours of total daily sleep, ≤6 hours per day was associated with higher overall cancer incidence: HR = 1.27 (95% CI 1.01–1.61).
The association was stronger for cancers of the digestive and respiratory tracts: HR = 1.41 (95% CI 1.03–1.93) for short total sleep vs 6–9 hours.
Long total sleep (>9 hours) showed a trend toward higher cancer risk, but estimates were less stable and not consistently significant across models.
In participants ≤60 years old, short total sleep was linked to a 35% higher overall cancer risk and an 83% higher risk for digestive and respiratory cancers; these age-stratified effects were weaker and less precise in those above 60.
The association held up after adjustment for a long list of confounders, including BMI, metabolic health, and lifestyle habits.
Study 4: What animal work shows about light, and glioblastoma growth
A 2025 experimental study—used a rat model to examine how constant light exposure affect glioblastoma (a brain tumor) growth and tumor biology. (PMC)
Rats were divided into several groups: normal light–dark cycles, constant light, light–dark with tumor, constant light with tumor.
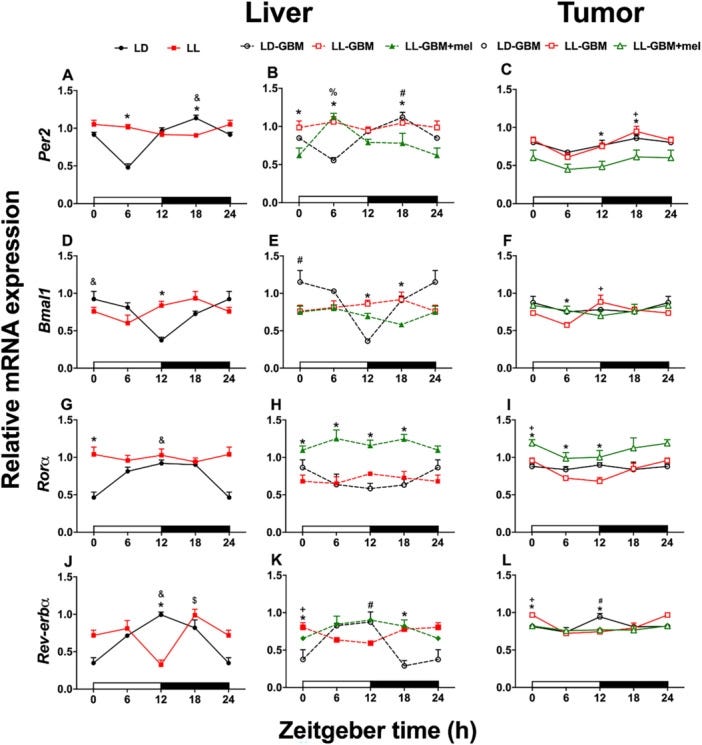
Tumor cells were injected, and the researchers tracked locomotor activity, body temperature, plasma melatonin rhythms, tumor volume, tumor weight, and expression of clock genes and angiogenesis genes (Angiogenesis refers to the growth of new blood vessels that may supply tumors.)
Key findings for tumor biology and circadian rhythms:
Constant light disrupted 24-hour patterns of locomotor activity and body temperature.
Rats with glioblastoma under constant light developed significantly larger tumors than tumor-bearing rats kept on normal light–dark cycles, with separation in tumor growth curves emerging ~13 days after tumor injection.
Constant light altered the rhythms of clock genes (Per2, Bmal1, Rorα, Rev-erbα) in liver and tumor tissue.
Constant light also changed the rhythmic expression of pro-angiogenic genes (VEGF-A, PDGF-C, TNF-α, Ang).
This is an animal study, so we can’t directly map these numbers onto human risk.
But it offers a mechanistic story that fits the human epidemiology: intense light exposure during what should be your biological night disrupts circadian timing in both healthy tissues and tumors, and speeds tumor growth.
The takeaway is that protecting darkness and circadian alignment at nighttime is not just about feeling rested. That is relevant if you often work or use screens late into the evening hours under bright light.
Five actions you can take
Drawing these pieces together, here are five practical moves that are grounded in the studies above and in surrounding evidence.
1. Aim for ~7-8 hours of total sleep per 24 hours, and avoid chronic ≤6 hours if you can.
The CHARLS cohort showed a 27% higher overall cancer risk and 41% higher digestive/respiratory cancer risk in people sleeping ≤6 hours per 24 hours compared with 6–9 hours.
The NHANES survivor study found both short and long sleep linked to higher cancer mortality vs 6–8 hours.
For you, this means tracking your total sleep time (not just time in bed) over weeks and trying to keep your typical band between ~7-8 hours unless a specialist has advised otherwise.
2. Nudge your schedule earlier and stabilize your sleep–wake timing.
The 2026 meta-analysis indicated that evening chronotype carries about 12–15% higher risk for some reproductive cancers vs morning chronotype, pointing to circadian misalignment as a contributor.
You don’t need to a large change at once. Instead, move your schedule by 15–30 minutes every few days: morning light exposure soon after waking, earlier last meal, and earlier wind-down, so your sleep midpoint gradually moves earlier and becomes more stable across the week. Even partial steps that make your weekdays and weekends look more alike can make a difference.
3. If you or someone you care about is a cancer survivor, combine mid-range sleep with regular moderate-to-vigorous activity.
In the NHANES analysis, survivors with short or long sleep who did not meet MVPA guidelines had the highest mortality; survivors who both met MVPA targets and had mid-range sleep did markedly better.
For you, that looks like: aiming for 7–8 hours of sleep plus at least 600 MET-minutes/week (for example, 150 minutes of brisk walking or equivalent) unless your physician has given different advice. If you already have limitations, you can still push activity as far as is safe and coordinate with your clinicians so sleep and movement plans fit your situation.
4. Protect darkness at nighttime.
The rat glioblastoma experiment showed that constant light flattened melatonin rhythms, sped tumor growth, and altered tumor blood vessels.
In human terms, the strategic move is not to supplement melatonin without cause but to protect your own melatonin production: keep your bedroom dark (including blackout curtains if needed), use dimmer, warmer light for at least an hour before bed, limit ceiling-level lighting in the late evening, and avoid bright screens close to your face late in your sleep window.
5. Address chronic insomnia and sleep-disordered breathing.
The 2026 review found higher breast cancer risk in people with insomnia in less adjusted models, and although the association weakened with better adjustment, it fits a body of work linking chronic sleep disturbance to higher inflammation, altered sex hormones, and metabolic risk—which all feed into cancer pathways.
If you have trouble staying asleep, long sleep-onset latency, or non-restorative sleep most nights of the week, or symptoms of sleep apnea (loud snoring, gasping, witnessed pauses), it is reasonable to treat those as longevity related improvements. Cognitive behavioral therapy for insomnia, proper evaluation for sleep apnea, and targeted support can help you move toward the mid-range sleep patterns that show up as beneficial for overall longevity.
Sleep is a modifiable factor to tilt the terrain of cancer risk
For someone in midlife and beyond, “good sleep” in this context means: enough hours across the 24-hour day, reasonably consistent timing that respects the light–dark cycle, and a dark, melatonin-friendly nighttime environment. The human cohorts suggest that keeping your total sleep in the moderate band and avoiding highly irregular patterns may modestly lower your odds of developing certain cancers, especially when you pair sleep with movement, non-smoking, and metabolic control.
If you treat sleep as one of your main health practices alongside nutrition and exercise, you stack benefits: better immune function, more resilient mood, steadier energy, and a more cancer-resistant internal environment over the coming years.
You’re building a sleep pattern that supports the brain, the immune network, and the tissues that need to repair themselves while you sleep.
Warmly,
—Kat
P.S. If you’ve tried the ideas in this email and you’re still stuck at 5–6 hours a night, I help people sort through the next steps here.
References
Pagkalidou, E., Papagiannopoulos, C. K., Manou, M., Filis, P., Richmond, R. C., Tsilidis, K. K., et al. (2026). Unravelling the association between sleep traits and reproductive cancers: A systematic review and meta-analysis. Sleep Medicine Reviews. https://www.sciencedirect.com/science/article/pii/S1087079226000079
Chen, R., Han, J., Li, S., Deng, H., Jian, T., Huang, Z., Wei, Y., & Fu, Z. (2025). Interaction between sleep duration and physical activity on mortality among cancer survivors: findings from National Health and Nutrition Examination Surveys 2007–2018. Frontiers in Public Health. https://www.frontiersin.org/articles/10.3389/fpubh.2025.1532320
Jiang, Y., Gu, X., Yang, X., Sun, A., & Sun, H. (2024). Exploring the association between sleep duration and cancer risk in middle-aged and older Chinese adults: observations from a representative cohort study (2011–2020). BMC Public Health, 24, 1819. https://link.springer.com/article/10.1186/s12889-024-19313-z
Cárdenas-Romero, M. L., Osnaya-Ruiz, A., Catalán, A., Ramírez-Plascencia, O. D., Guerrero-Velázquez, C., Ángeles-Casas, L. G., et al. (2025). Melatonin prevents tumor growth: The role of genes controlling the circadian clock, the cell cycle, and angiogenesis. Journal of Pineal Research, 77(4), e70064. https://pmc.ncbi.nlm.nih.gov/articles/PMC12167844/